Uranium
In nature, uranium is found as uranium-238 (99.2739–99.2752%), uranium-235 (0.7198–0.7202%), and a very small amount of uranium-234 (0.0050–0.0059%). Uranium decays slowly by emitting an alpha particle. The half-life of uranium-238 is about 4.47 billion years and that of uranium-235 is 704 million years making them useful in dating the age of the Earth.
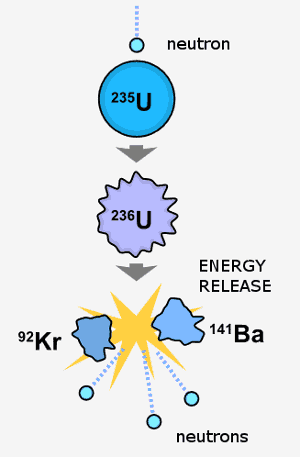
Many contemporary uses of uranium exploit its unique nuclear properties. Uranium-235 has the distinction of being the only naturally occurring fissile isotope. Uranium-238 is fissionable by fast neutrons, and is fertile, meaning it can be transmuted to fissile plutonium-239 in a nuclear reactor. Another fissile isotope, uranium-233, can be produced from natural thorium and is also important in nuclear technology. While uranium-238 has a small probability for spontaneous fission or even induced fission with fast neutrons, uranium-235 and to a lesser degree uranium-233 have a much higher fission cross-section for slow neutrons. In sufficient concentration, these isotopes maintain a sustained nuclear chain reaction. This generates the heat in nuclear power reactors, and produces the fissile material for nuclear weapons. Depleted uranium (238U) is used in kinetic energy penetrators and armor plating.
Uranium is used as a colorant in uranium glass, producing orange-red to lemon yellow hues. It was also used for tinting and shading in early photography. The 1789 discovery of uranium in the mineral pitchblende is credited to Martin Heinrich Klaproth, who named the new element after the planet Uranus. Eugène-Melchior Péligot was the first person to isolate the metal and its radioactive properties were discovered in 1896 by Antoine Becquerel. Research by Enrico Fermi and others starting in 1934 led to its use as a fuel in the nuclear power industry and in Little Boy, the first nuclear weapon used in war. An ensuing arms race during the Cold War between the United States and the Soviet Union produced tens of thousands of nuclear weapons that used uranium metal and uranium-derived plutonium-239. The security of those weapons and their fissile material following the breakup of the Soviet Union in 1991 is an ongoing concern for public health and safety. See Nuclear proliferation.
Occurrence
Biotic and abiotic
Uranium is a naturally occurring element that can be found in low levels within all rock, soil, and water. Uranium is the 51st element in order of abundance in the Earth's crust. Uranium is also the highest-numbered element to be found naturally in significant quantities on Earth and. Along with all elements having atomic weights higher than that of iron, it is only naturally formed in supernovae. The decay of uranium, thorium, and potassium-40 in the Earth's mantle is thought to be the main source of heat that keeps the outer core liquid and drives mantle convection, which in turn drives plate tectonics.
Uranium's average concentration in the Earth's crust is (depending on the reference) 2 to 4 parts per million, or about 40 times as abundant as silver. The Earth's crust from the surface to 25 km (15 mi) down is calculated to contain 1017 kg (2×1017 lb) of uranium while the oceans may contain 1013 kg (2×1013 lb). The concentration of uranium in soil ranges from 0.7 to 11 parts per million (up to 15 parts per million in farmland soil due to use of phosphate fertilizers), and its concentration in sea water is 3 parts per billion.
Uranium is more plentiful than antimony, tin, cadmium, mercury, or silver, and it is about as abundant as arsenic or molybdenum. Uranium is found in hundreds of minerals including uraninite (the most common uranium ore), carnotite, autunite, uranophane, torbernite, and coffinite. Significant concentrations of uranium occur in some substances such as phosphate rock deposits, and minerals such as lignite, and monazite sands in uranium-rich ores (it is recovered commercially from sources with as little as 0.1% uranium).Some bacteria such as S. putrefaciens and G. metallireducens have been shown to reduce U(VI) to U(IV).
Some organisms, such as the lichen Trapelia involuta or microorganisms such as the bacterium Citrobacter, can absorb concentrations of uranium that are up to 300 times higher than in their environment.Citrobacter species absorb uranyl ions when given glycerol phosphate (or other similar organic phosphates). After one day, one gram of bacteria can encrust themselves with nine grams of uranyl phosphate crystals; this creates the possibility that these organisms could be used in bioremediation to decontaminate uranium-polluted water.
In nature, uranium(VI) forms highly soluble carbonate complexes at alkaline pH. This leads to an increase in mobility and availability of uranium to groundwater and soil from nuclear wastes which leads to health hazards. However, it is difficult to precipitate uranium as phosphate in the presence of excess carbonate at alkaline pH. A Sphingomonas sp. strain BSAR-1 has been found to express a high activity alkaline phosphatase (PhoK) that has been applied for bioprecipitation of uranium as uranyl phosphate species from alkaline solutions. The precipitation ability was enhanced by overexpressing PhoK protein in E. coli.
Plants absorb some uranium from soil. Dry weight concentrations of uranium in plants range from 5 to 60 parts per billion, and ash from burnt wood can have concentrations up to 4 parts per million. Dry weight concentrations of uranium in food plants are typically lower with one to two micrograms per day ingested through the food people eat.
Production and mining
The worldwide production of uranium in 2010 amounted to 53,663 tonnes, of which 17,803 t (33.2%) was mined in Kazakhstan. Other important uranium mining countries are Canada (9.783 t), Australia (5,900 t), Namibia (4,496 t), Niger (4,198 t) and Russia (3,562 t).
Uranium ore is mined in several ways: by open pit, underground, in-situ leaching, and borehole mining (see uranium mining). Low-grade uranium ore mined typically contains 0.01 to 0.25% uranium oxides. Extensive measures must be employed to extract the metal from its ore. High-grade ores found in Athabasca Basin deposits in Saskatchewan, Canada can contain up to 23% uranium oxides on average. Uranium ore is crushed and rendered into a fine powder and then leached with either an acid or alkali. The leachate is subjected to one of several sequences of precipitation, solvent extraction, and ion exchange. The resulting mixture, called yellowcake, contains at least 75% uranium oxides U3O8. Yellowcake is then calcined to remove impurities from the milling process before refining and conversion.
Commercial-grade uranium can be produced through the reduction of uranium halides with alkali or alkaline earth metals. Uranium metal can also be prepared through electrolysis of KUF5 or UF4, dissolved in molten calcium chloride (CaCl2) and sodium chloride (NaCl) solution. Very pure uranium is produced through the thermal decomposition of uranium halides on a hot filament.
Resources and reserves
It is estimated that 5.5 million tonnes of uranium exists in ore reserves that are economically viable at US$59 per lb of uranium, while 35 million tonnes are classed as mineral resources (reasonable prospects for eventual economic extraction). Prices went from about $10/lb in May 2003 to $138/lb in July 2007. This has caused a big increase in spending on exploration, with US$200 million being spent world wide in 2005, a 54% increase on the previous year,. This trend continued through 2006, when expenditure on exploration rocketed to over $774 million, an increase of over 250% compared to 2004. The OECD Nuclear Energy Agency said exploration figures for 2007 would likely match those for 2006.
Australia has 31% of the world's known uranium ore reserves and the world's largest single uranium deposit, located at the Olympic Dam Mine in South Australia. There is a significant reserve of uranium in Bakouma a sub-prefecture in the prefecture of Mbomou in Central African Republic.
Some nuclear fuel comes from nuclear weapons being dismantled, such as from the Megatons to Megawatts Program.
An additional 4.6 billion tonnes of uranium are estimated to be in sea water (Japanese scientists in the 1980s showed that extraction of uranium from sea water using ion exchangers was technically feasible).There have been experiments to extract uranium from sea water, but the yield has been low due to the carbonate present in the water. In 2012, ORNL researchers announced the successful development of a new absorbent material dubbed HiCap, which vastly outperforms previous best adsorbents, which perform surface retention of solid or gas molecules, atoms or ions. "We have shown that our adsorbents can extract five to seven times more uranium at uptake rates seven times faster than the world's best adsorbents," said Chris Janke, one of the inventors and a member of ORNL's Materials Science and Technology Division. HiCap also effectively removes toxic metals from water, according to results verified by researchers at Pacific Northwest National Laboratory.
Supply
In 2005, seventeen countries produced concentrated uranium oxides, with Canada (27.9% of world production) and Australia (22.8%) being the largest producers and Kazakhstan (10.5%), Russia (8.0%), Namibia (7.5%), Niger (7.4%), Uzbekistan (5.5%), the United States (2.5%), Argentina (2.1%), Ukraine (1.9%) and China (1.7%) also producing significant amounts. Kazakhstan continues to increase production and may have become the world's largest producer of uranium by 2009 with an expected production of 12,826 tonnes, compared to Canada with 11,100 t and Australia with 9,430 t. The ultimate available uranium is believed to be sufficient for at least the next 85 years, although some studies indicate underinvestment in the late twentieth century may produce supply problems in the 21st century. Uranium deposits seem to be log-normal distributed. There is a 300-fold increase in the amount of uranium recoverable for each tenfold decrease in ore grade. In other words, there is little high grade ore and proportionately much more low grade ore available.
| Symbol | U | |
| Atomic Number | 92 | |
| Atomic Weight | 238.0289 | |
| Oxidation States | +3, +4, +5, +6 | |
| Electronegativity, Pauling | 1.36 | |
| State at RT | Solid, Metal | |
| Melting Point, K | 1405.5 | |
| Boiling Point, K | 4018 |
Appearance and Characteristics
Harmful effects:
Uranium is harmful both through its chemical toxicity and its radioactivity. Exposure to uranium increases your risk of getting a variety of cancers due to its radioactivity.
Characteristics:
- Uranium is a dense, silvery-white, slightly paramagnetic, radioactive metal. It is also ductile and malleable. The metal tarnishes in air acquiring a dark layer of oxide. When finely powdered, uranium ignites spontaneously in air.
- Uranium is a highly reactive metal and reacts with almost of all the nonmetallic elements and many of their compounds. It dissolves in acids, but it is insoluble in alkalis.
- All isotopes of uranium are radioactive, some more so than others. Its radioactivity – in particular its capacity to undergo thermonuclear chain reactions – has led to uranium’s use in energy generation, both for civilian and military purposes.
Uses of Uranium
- Uranium is used as fuel for nuclear power plants. One kilogram of uranium-235 has the capacity to produce as much energy as 1,500,000 kilograms (1,500 tonnes) of coal.
- Naturally occurring uranium is over 99% U-238 with only about 0.7% of the fissile U-235. Huge gas diffusion plants are used to produce enriched uranium, which has higher concentrations of U-235.
- Uranium for use in nuclear power plants is enriched to a U-235 concentration of 2-3%.
- In nuclear weapons, it is believed uranium is enriched to about 90% U-235, although lower concentrations would still yield a working bomb.
- Depleted uranium is a byproduct of enriching uranium for nuclear purposes. It contains about 0.2% U-235 and is about half as radioactive as naturally occurring uranium.
- Its lower radioactivity has allowed depleted uranium to be used in applications where uranium’s very high density is useful. (A tennis ball sized sphere of uranium would weigh about 5.7 pounds (2.6 kilograms).) It is used by the military as shielding to protect army tanks, and also in parts of bullets and missiles.
- Use of depleted uranium in missiles is controversial because, on impact, uranium vapor and dust form and these are highly toxic.
- U-238 can be converted into fissionable plutonium in breeder reactors.